|
Magnetic Resonance Imaging |
|
 |
Magnetic resonance imaging (MRI) is one of
the most commonly used tests in neurology and neurosurgery. MRI provides
exquisite detail of brain, spinal cord and vascular anatomy. MRI has the
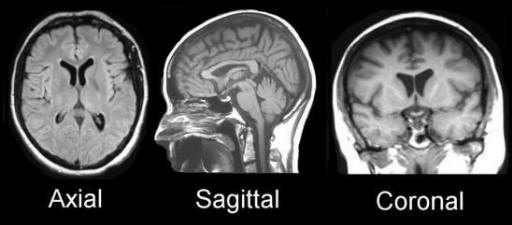
advantage of being able to visualize anatomy in all three planes: sagittal,
axial and coronal. MRI can detect flowing blood and cryptic vascular
malformations. It can also detect demyelinating disease, and has no
beam-hardening artifacts such as can
be seen with CT. This results in the the posterior fossa being more easily
visualized on MRI than CT. Imaging is also performed without any ionizing radiation.
|
|
|
PHYSICS OF MRI
MRI is based on the magnetization properties of atomic nuclei. A powerful,
uniform, external magnetic field is employed to align the normally randomly
oriented protons within water nuclei in the tissue being examined. This
alignment (or magnetization) is next perturbed or disrupted by introduction of
an external Radio Frequency (RF) energy. The nuclei return to their resting
alignment through various relaxation processes and in so doing emit RF energy.
After an appropriate period following the initial RF, the emitted signals are
measured. Fourier transformation is used to convert the frequency information
contained in the signal from each location in the imaged plane to corresponding
intensity levels, which are then displayed as shades of gray in a matrix
arrangement of pixels. By varying the sequence of RF pulses applied & collected,
different types of images are created. REPETITION TIME
(TR) is the amount of time between successive pulse sequences applied
to the same slice. TE (Echo Time) -
represents the time between the delivery of the RF pulse and the receipt of the
echo signal.
Tissue can be characterized by two kinds of relaxation times – T1 and T2.
T1 - LONGITUDINAL RELAXATION TIME – is the time
constant which determines the rate at which excited protons return to
equilibrium. It is a measure of the time taken for spinning protons to re-align
with the external magnetic field. T2 – TRANSVERSE
RELAXATION TIME – is the time constant which determines the rate at
which excited protons reach equilibrium, or go out of phase with each other. It
is a measure of the time taken for spinning protons to lose phase coherence
among the nuclei spinning perpendicular to the main field.
|
MRI IMAGING SEQUENCES
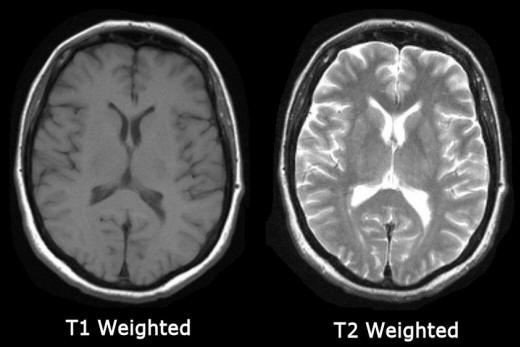
T1 weighted images are produced by using
short TE and TR times. The contrast and brightness of the image are
predominately determined by T1 properties of tissue. Conversely,
T2 weighted imaging is produced by using longer
TE and TR times. In these images, the contrast and brightness are predominately
determined by the T2 properties of tissue.
|
|
|
In general, T1 and T2 weighted images can be easily differentiated by looking
the CSF. CSF is dark on T1 weighted imaging and bright on T2 weighted imaging (see
figure above).
|
|

|
|
Table Above: Common tissues and pathology
on T1 and T2 weighted imaging.
T1-weighted imaging can also be performed while infusing
GADOLINIUM (Gad). Gad is a non-toxic
paramagnetic contrast enhancement agent. When injected during the scan, Gad will
tend to change signal intensities by shortening T1. Thus, Gad is very bright on
T1 weighted images. Gad enhanced images are especially useful in looking at
vascular structures and breakdown in the blood brain barrier [e.g., tumors,
abscess, inflammation (herpes simplex encephalitis, multiple sclerosis, etc.)].
|
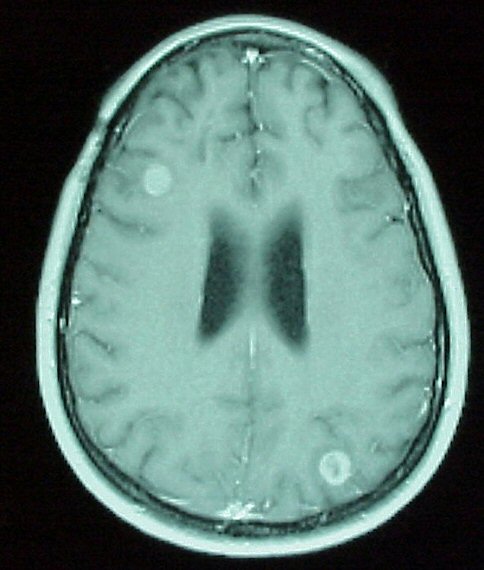 |
Left:
Axial MRI with Gad. Note the enhancing lesions in the right frontal and left
occipital areas, consistent with metastatic disease. On routine T1 imaging,
these lesions were not visible. |
Diffusion weighted imaging (DWI) is
designed to detect the random movements of water protons. Water molecules
diffuse relatively freely in the extracellular space; their movement is
significantly restricted in the intracellular space. Spontaneous movements,
referred to as diffusion, rapidly become restricted in ischemic brain tissue.
During ischemia, the Na/K pump shuts down and Na+ accumulates intracellularly.
Water then shifts from the extracellular to the intracellular space due to the
osmotic gradient. As water movement becomes restricted intracellularly, this results in an extremely bright signal on DWI.
Thus, DWI has become an extremely sensitive method in the detection of acute
stroke.
|
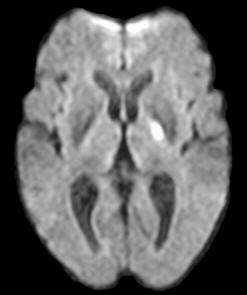 |
Left:
Diffusion weighted imaging. Note the obvious lesion in the left posterior limb
of the internal capsule. Routine T1 and T2 weighted imaging was normal. |
NEUROLOGIC INDICATIONS FOR CRANIAL MRI
Vascular
Ischemic and hemorrhagic
stroke; AVM, aneurysm, venous thrombosis
Tumor
Primary CNS and metastatic
Infection
Abscess, cerebritis,
encephalitis, meningitis
Inflammatory Lesions
Multiple sclerosis, sarcoidosis,
etc.
Trauma
Epidural hematoma, subdural
hematoma, contusion
Hydrocephalus
Congenital Malformations
|
LIMITATIONS OF MRI
Subject to motion artifact
Inferior to CT in detecting
acute hemorrhage
Inferior to CT in detection of
bony injury
Requires prolonged acquisition
time for many images |
CONTRAINDICATIONS TO MRI
There are few contraindications to MRI. Conventional MRI in patients able to
undergo the procedure have no associated biological effects. Most
contraindications to MRI are can be divided into the following groups classes:
• Implanted devices and other metallic
devices
Pacemakers and other implanted
electronic devices
Aneurysm clips and other
magnetizable materials
Cochlear implants
Some artificial heart valves
• Intraocular metallic foreign bodies
Screening CT of the orbits if
history suggests possible metallic
foreign body in the eye
• Unstable patients
Most resuscitation
equipment cannot be brought into the scanning room
• Pregnancy
Relative contraindication due
to unknown effects on fetus
• Other – severe agitation, or claustrophobia
(may require anesthesia assistance) |